New groundbreaking discovery found that recharging specific eye cells can prevent total blindness
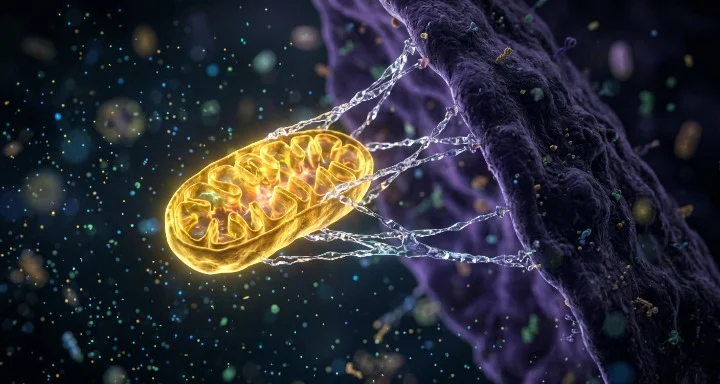
Imagine the world slowly going dark. It isn’t a sudden snap of the fingers; it is a creeping grayness starting at the center of your vision and eating its way outward. For patients with Leber’s hereditary optic neuropathy (LHON), this isn’t a metaphor. It is a biological death sentence for the cells in the optic nerve. The culprit is a tiny, ancient glitch in their DNA—specifically, a mutation that causes their mitochondria to stop producing the energy required to keep a neuron alive. Without power, the cells simply stop working, and eventually, they vanish.
For decades, modern medicine has been largely powerless against these mitochondrial diseases. We could treat the symptoms, but the fundamental problem—the broken “batteries” inside the cell—remained out of reach. That changed this week.
According to a study published in Nature on April 15, 2026, a team led by Temurkhan Ayupov and Botond Roska has developed a system they call MitoCatch. It is a method for delivering fresh, healthy mitochondria specifically to the cells that need them most, effectively performing a biological battery swap in a living organism.
The Postal Code of the Cell
The problem with mitochondrial transplantation has never been the “what.” Scientists have known for years that you can isolate healthy mitochondria from one cell and put them near another. The problem is the “how” and the “where”. Cells are picky. If you just dump mitochondria into the bloodstream, they don’t know where to go. They end up everywhere except the specific neurons or heart cells that are actually dying.
The MitoCatch system solves this by using what are essentially biological “postage stamps”. The researchers looked at how viruses enter cells. Viruses use specific proteins on their coat to latch onto receptors on a cell’s surface. The team decided to mimic this trick. By engineering protein binders—specifically nanobodies and antibodies—they created a bridge. One end of the bridge sticks to the outer membrane of a healthy donor mitochondrion; the other end is programmed to find a specific “address” on the target cell, such as a neuron in the retina or a muscle cell in the heart.
In their tests, the results were almost unbelievable. When they used these binders, the targeted cells saw an enrichment of healthy mitochondria that was 1,300% higher than cells without the “postal code”.
More Than Just a Guest
It is one thing to stick a healthy organelle to the outside of a cell; it is quite another to get the cell to accept it. The researchers watched under high-resolution microscopes as these donor mitochondria were swallowed by the target cells and released into the cytosol.
But do they actually work? The team didn’t just want to see mitochondria sitting inside a cell like a piece of furniture. They wanted to see them join the team. Using live imaging, they watched as the transplanted mitochondria began to move along the long, spindly branches of neurons. They even observed the donor mitochondria undergoing “fusion and fission”—the biological equivalent of merging and splitting to maintain health—with the cell’s native, failing mitochondria.
In LHON patient-derived cells, the effect was profound. These cells were starving for energy, and their survival rates were plummeting. Once the fresh mitochondria were “delivered,” the cells upregulated their energy production. Their respiration rates surged, and their survival increased significantly. The new “batteries” weren’t just guests; they had taken over the power grid.
The Return of Sight
The most gripping part of the research happened when the team moved from the lab dish to the living eye. In mice with optic nerve injuries—a stand-in for the damage seen in human glaucoma or neurodegeneration—the researchers injected healthy mitochondria tagged with the MitoCatch system.
The healthy organelles didn’t just linger at the injection site. They tracked down the injured ganglion cells and moved inside. Ten days later, the mice that received the “energy mail” had a 46.8% higher survival rate of their vision-critical neurons compared to those that were left untreated.
The team even went a step further, measuring the actual light responses in the mice’s brains. They found that the treated retinas were actively responding to light stimuli again. They weren’t just saving cells; they were saving function. The mice could “see” in ways the untreated group simply could not.
The Frontier of Organelle Therapy
We are looking at the birth of a new category of medicine: organelle therapy. This isn’t gene therapy, where we try to rewrite the code; it is a direct replacement of the hardware.
The implications for “untreatable” conditions are massive. Heart failure, which is often a result of energy-starved cardiac muscle, could be treated with a localized injection of healthy mitochondria. Immune cells could be “supercharged” for better cancer fighting. And for the millions of people facing slow-motion blindness from mitochondrial decay, there is finally a reason to believe the lights won’t go out.
There is still work to do, of course. We need to know how long these “transplants” last and whether the body’s immune system will eventually notice the new guests. But for now, the MitoCatch system has proven that the “energy gap” in human disease isn’t an insurmountable wall. It’s just a delivery problem. And we finally have the right address.